Icephobic materials development and testing
Danish Technological Institute (DTI) from Denmark, Industrielack AG (ILAG) – a Swiss company and the Refrigeration Institute from Karlsruhe University of Applied Sciences (UASKA), together with SPF Institute for Solar Technology (SPF) are investigating a large variety of icephobic coatings. The aim is to identify a durable icephobic coating for developing a compact and reliable supercooler (evaporator). The evaporator will be then used for the new heat pump from the TRI-HP solar ice-slurry system.
Icephobic coatings and testing facility for evaluating the underwater icing process at DTI (Denmark)
DTI develops coatings to prevent/delay the ice formation on the walls of supercooling heat exchangers. By applying coatings, the risk of ice formation in the heat exchanger is minimized and the water can be supercooled. DTI's coatings are isocyanate free coatings, using hybrid silicone-epoxy resins as main coating constituents.

While a large number of icephobic materials was reported for air applications (e.g. for aerospace industry), data on underwater icephobic materials and their performance characterization can be hardly found. DTI has therefore developed a special test rig for the TRI-HP project to evaluate the underwater icing process on lab scale samples.
The testing chamber is made of stainless steel and has two main parts (see image below). A cooling block consisting of a Peltier element and a thermocouple is situated in the lower part to cool the test sample from the backside. The top part mounted over the lower part has a cylindrical opening that serves as water tank. An external agitator is used to induce flow dynamics and two USB microscopes permit ice formation monitoring.

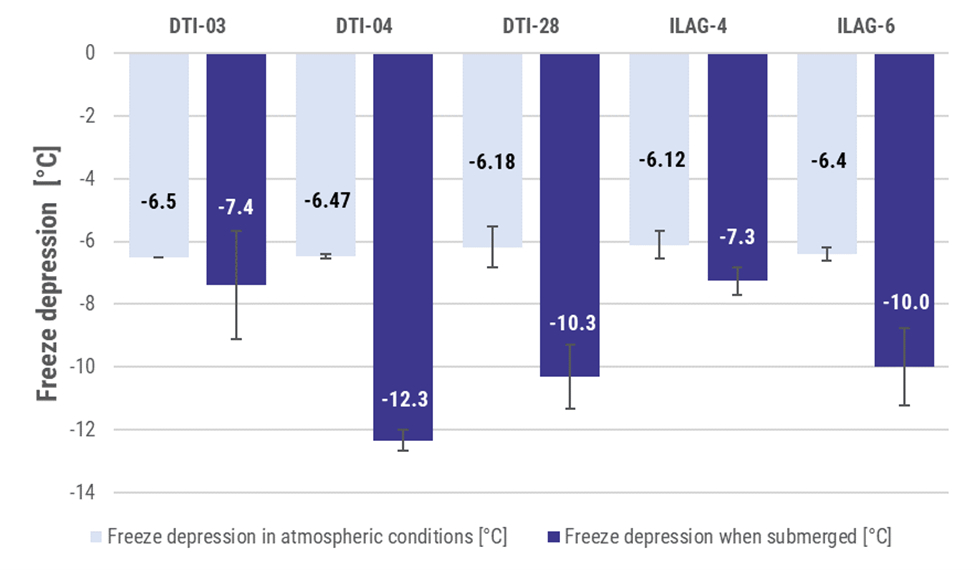
Results at sample level with the new testing device (submerged samples), as well as in atmospheric conditions are presented in the graph below. They indicate that several coatings can effectively prevent the formation of ice and promote a higher supercooling degree compared to uncoated references. Subsequently, both DTI and ILAG coatings were sprayed on copper and stainless-steel tubes to be validated at SPF in a heat exchanger like environment. Currently, real coated heat exchanger are being tested in a similar manner at SPF laboratories.
Bulk amorphous materials testing at UASKA
At UASKA different solutions to enable reliable supercooling of water are investigated.
Freeze depression tests carried out at UASKA prior to the TRI-HP project have shown that materials with a different crystal structure than that of an ice particle were able to achieve a higher supercooling compared to surface with structures similar to ice crystals. Thus, amorphous and nano-crystalline materials were tested first since their structure theoretically inhibits the ice formation.
A preliminary investigation was carried out applying water droplets on three different amorphous alloys. Each sample material achieved a supercooling of at least -8 °C. Besides the freeze depression, a freeze delay test was performed. Even after two hours at a temperature of -8 °C, the water remained liquid.
A variety of ten amorphous and nano-crystalline metals was then tested to investigate possible influencing effects of the alloying elements (table below) before implementing them in the test rig.
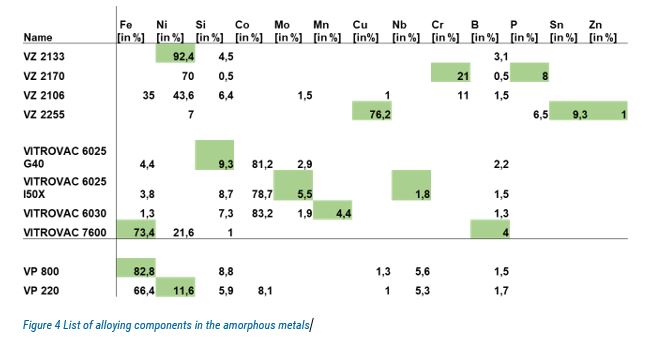
In contrast to conventional metals, such as aluminium and copper, the tested materials were able to reach the final temperature of -11 °C and maintain it for two hours or longer without water droplet freezing. The results show an increase in supercooling when nickel is present in the alloy. However, none of the alloys showed good durability when immersed in water. In most cases, degradation occurred quite soon.